Study shows mpox vaccine generates immune response to emerging clade Ib, but protection levels remain unclear
April 15, 2026

April 15, 2026
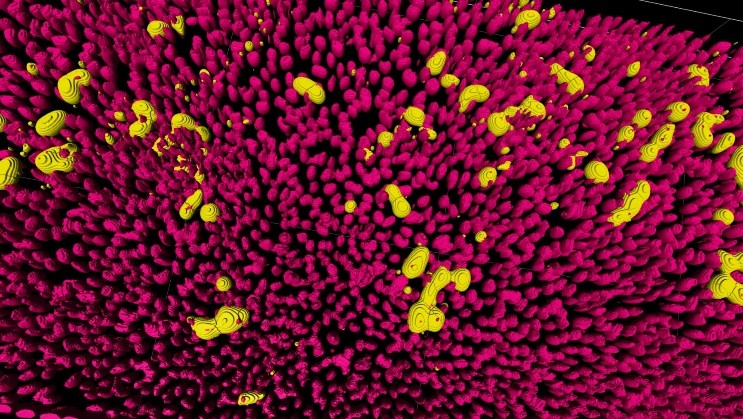
New research has identified key mutations in the Middle East respiratory syndrome coronavirus (MERS-CoV) that may influence how easily the virus infects cells and evades immune responses, offering important insights for pandemic preparedness. The study, supported by funding from The Pandemic Institute and led by researchers at the University of Liverpool plus colleagues from across…

The Pandemic Institute Public Steering Group recently met for its fifth session, bringing together community members and researchers to review ongoing work and discuss a new study focused on vaccine inequalities. The meeting opened with updates from the Institute, including progress on two research projects: an AI-assisted robotic microscope being used in a high-containment laboratory…